1/2/19 · Baking soda and vinegar have an acidbased reaction, which results in the gas that fills the balloon Baking Soda and vinegar react chemically we know that much simply by observing the experiments We can see the chemical reactionThe balloon will fill up from the gases produced from the chemical reaction between the baking soda and vinegar the reaction that takes in between the two compounds leads to the evolution of carbon dioxide gas which infiltrates the balloon and blows up You just need to be able to seal the end of the balloon around a topWhen vinegar and baking soda mix, they create the gas carbon dioxide and water The carbon dioxide has no where to go, but into the balloon – blowing it upThis is because the colder temperature causes the reaction to take place more slowly, releasing the gas less effectively

Balloon Baking Soda Vinegar Science Experiment For Kids
Vinegar and baking soda chemical reaction balloon
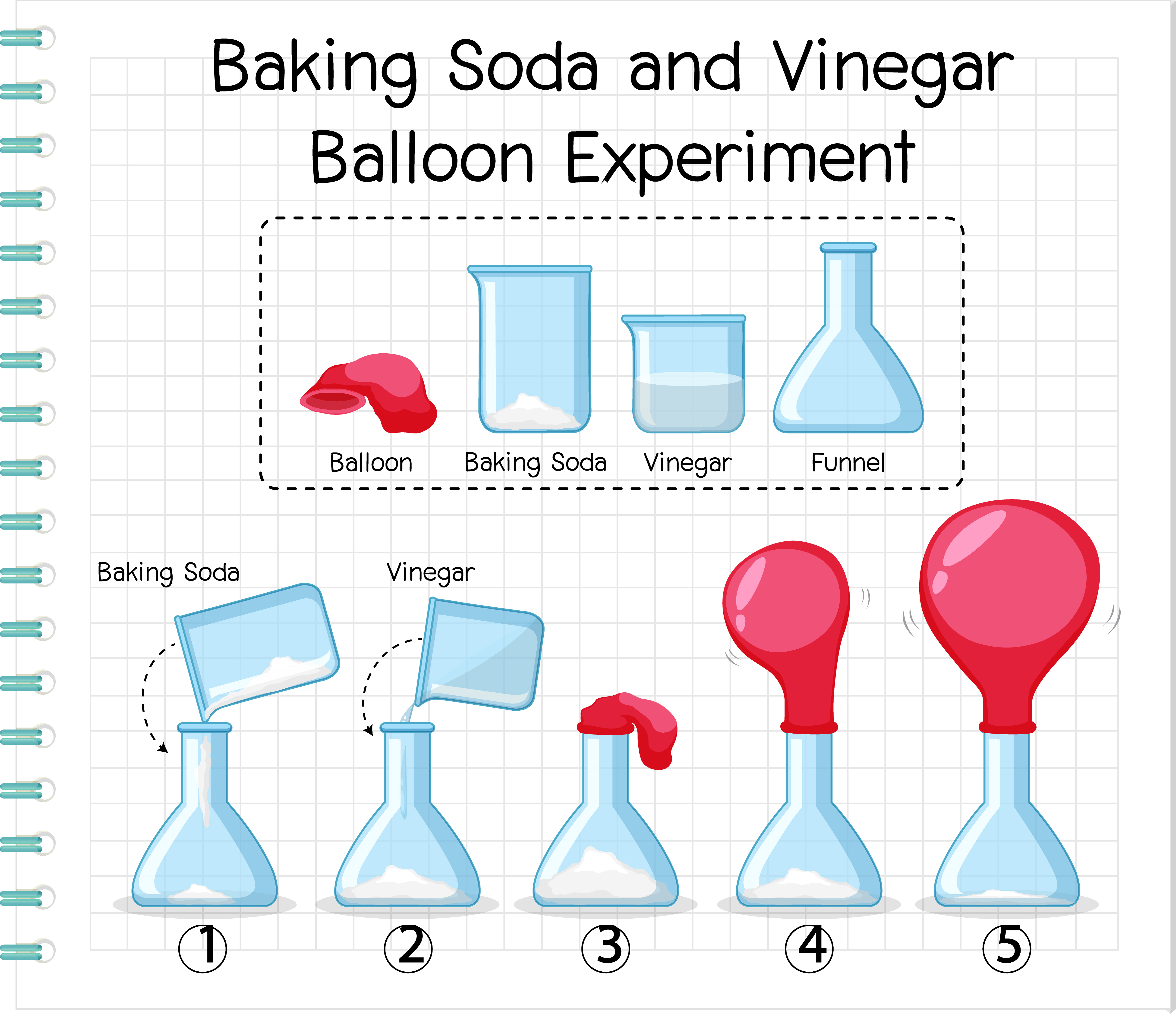
Vinegar and baking soda chemical reaction balloon-19/3/19 · Baking Soda And Vinegar Balloon Lab Report Science laboratory reports are created to communicate the findings of research study, in such a way that is clear to readers You need to not forget to include any additional details, which might be beneficial for readers Composing a good science laboratory sample is vital if you wish to make your research and your reportBlow up balloon with baking soda and vinegar When vinegar is mixed with baking soda, a double replacement reaction takes place The end result is carbon dioxide but behind the scenes, there is more than one reaction taking place Put a tablespoon of backing soda on to the center of a



Magic Balloons Playdough To Plato
It's quite simple, combining vinegar and baking soda together is considered an acid base reaction Vinegar is acidic and baking soda is a base When you mix and acid with a base, two new chemicals form, carbonic acid and sodium acetateCarefully place the balloon over the top of the vinegar bottle without letting any of the baking sodaThis lab demonstrates the reactivity of two household cooking items, baking soda and vinegar Baking soda is a powdered chemical compound called sodium bicarbonate, and vinegar includes acetic acid These 2 components react in solution to form carbon dioxide, water, and sodium acetate as shown in the chemical reaction below
31/1/ · The overall chemical reaction between baking soda (sodium bicarbonate) and vinegar (weak acetic acid) is one mole of solid sodium bicarbonate reacts with one mole of liquid acetic acid to produce one mole each of carbon dioxide gas, liquid water, sodium ions, and acetate ions The reaction proceeds in two stepsThe first reaction is the acidbase reaction When vinegar and baking soda are first mixed together, hydrogen ions in the vinegar react with the sodium and bicarbonate ions in the baking soda The result of this initial reaction is two new chemicals carbonic acid and sodium acetate14/12/19 · You have a party at home, but you feel lazy to empty your lungs out into all those balloons Your search for an easier way to blow up balloons ends here In the balloon vinegar baking soda experiment, you will learn about the reactions between vinegar and baking soda You will also learn about the gas produced when combining vinegar and baking
The reaction c SELFINFLATING BALLOON BAKING SODA AND VINEGAR HUNGRY SciANNtistThe reaction between baking soda and vinegar is called AcidBase reaction8/10/16 · Once the vinegar and baking soda mix, the balloon will start to automatically inflate For those inquisitive minds who want to know how this happens, the vinegar is an acid and baking soda is a base When you mix an acid with a base, it causes a chemical reactionThen, all of the balloons blew up completely so there was no difference between any of the balloons I decided to increase the amount of baking soda and vinegar by only 50% and this worked Measure 15 ml of baking soda using a measuring spoon Pour the baking soda into the balloon using a funnel Measure 45 ml of vinegar and pour it into a



Build A Fizz Inflator Sciencebob Com



Giant Balloon Baking Soda And Vinegar Experiment Hello Wonderful
Baking soda is bicarbonate (NaHCO3) and vinegar is acetic acid (HCH3COO) One of the products this reaction creates is carbon dioxide, which makes the bubbles When the baking soda meets the vinegar, there is a chemical reaction as carbon dioxide gas is created and fills the balloon causing it4/9/15 · Instructions Stretch the opening of the balloon over the end of the funnel Pour about 1/3 cup of baking soda into the funnel and Rinse all the baking soda off the funnel, and then use the funnel to pour the vinegar into a water bottle Gently stretch the opening of the balloon over the openingHow to Blow Up a Ballon with Baking Soda & Vinegar!


Easy Baking Soda Balloon Experiment



Magic Balloon Inflation Baking Soda And Vinegar Experiment Midwestern Mama
18/11/ · When vinegar and baking soda are first mixed together, hydrogen ions in the vinegar react with the sodium and bicarbonate ions in the baking soda The result of this initial reaction is two new chemicals carbonic acid and sodium acetate18/11/ · If the reaction is too weak to inflate the balloon, add more vinegar and baking soda , but don't shake the solution How do you blow up a balloon with a chemical reaction?The baking soda and vinegar chemical reaction makes sodium acetate, water, and carbon dioxide (photo Jinx!) The baking soda and vinegar chemical reaction finds use in chemical volcanoes, carbon dioxide production, and sodium acetate synthesisIt's an aqueous (waterbased) reaction between sodium bicarbonate and the acetic acid from vinegar


Science Experiments For Kids Blow Up A Balloon With Vinegar And Baking Soda


Science Experiment With Baking Soda And Vinegar Balloon Vector Art At Vecteezy
24/5/ · What is the chemical reaction between baking soda and vinegar that causes the balloon to blow up?8 Gently shake the balloon so the baking soda falls into the soda bottle to mix with the vinegar Keep the bottle still on the table 9 Observe what happens when the baking soda and vinegar mix Notice the temperature change that happens in the bottle as the reaction happens!1/2/17 · Step 4 Lift the balloon up over the bottle and watch the reaction The baking soda should fall out of the balloon, through the neck of the bottle, and into the vinegar at the bottom Here, the two chemicals will fizz and react, turning into other chemicals One of these is carbon dioxide, a gas, which will rise up and inflate the balloon



Build A Fizz Inflator Sciencebob Com



Balloon Baking Soda Vinegar Science Experiment For Kids
7/1/ · Baking soda is bicarbonate (NaHCO3) and vinegar is acetic acid (HCH3COO) One of the products this reaction creates is carbon dioxide, which makes the bubbles When the baking soda meets the vinegar, there is a chemical reaction as carbon dioxide gas is created and fills the balloon causing it to inflateTemperature amount of trails air pressure procedure hypothesis measure and record the circumference after the reaction 10 500ml water bottles balloons 2 colours funnel baking soda vinegar measuring cup ruler repeat steps 34 increasing 1 table spoon of baking soda MyThe science, behind this balloon baking soda experiment, is the chemical reaction between the base {baking soda} and the acid {vinegar} When the two ingredients mix together the balloon baking soda experiment gets it's lift!



Mom To 2 Posh Lil Divas Blow It Up Exploring Gas With Balloons Baking Soda Vinegar



Inflate A Balloon With Baking Soda And Vinegar Pbs Kids For Parents
The vinegar 7 Carefully stretch the open end of the balloon over the top of the open bottle 8 Turn on the black light and turn off the rest of the lights 9 Hold the bottle with one hand and carefully stand the balloon upright 10 Gently shake the balloon so the baking soda falls into the soda bottle to mix with the vinegar Keep the bottleExperiment Inflate a balloon with vinegar and baking soda reaction Baking soda and vinegar reaction inflate a gas balloon You can inflate a gas balloon without having to blow it itself What do you need a small glass bottle a funnel a balloon baking soda vinegar First add some vinegar into the bottle18/11/ · Baking soda is bicarbonate (NaHCO3) and vinegar is acetic acid (HCH3COO) One of the products this reaction creates is carbon dioxide, which makes the bubbles When the baking soda meets the vinegar, there is a chemical reaction as carbon dioxide gas is created and fills the balloon causing it to inflate



Balloon Magic With Baking Soda And Vinegar Gift Of Curiosity



Magic Balloons Playdough To Plato
10 When the reaction is complete, you can remove the balloon to28/4/ · Fun DIY Science you can do right in your kitchen at home!This is a very fun and easy science experiment for kids!!!Hey Everyone and welcome back to the labIn thi



Balloon Baking Soda Vinegar Science Experiment For Kids



Kid Science Hot Air Balloon Bottle Kix Cereal
Baking soda, vinegar, 12 balloons, small plastic bottle, spoon, funnel Procedures 1 Add two teaspoons of baking soda to a clear cup Observe closely 2 Add about 1/4 cup of vinegar to the cup Observe what happens 3 Pour about 1/4 cup of vinegar into a small empty plastic bottle 4 Add 2 teaspoons of baking soda to a balloon using aThe balloon should partially fill with gas Help your child remove and tie the balloon shortly after the fizzing stops This isn't your average balloon instead of blowing it up with her own breath or a fancy helium machine, your child has inflated a balloon with only baking soda and vinegar!Baking Soda Vinegar Balloon Experiment!



Baking Soda And Vinegar Balloons



Baymax S Low Battery Baking Soda And Vinegar Experiment
9/5/18 · First the vinegar will fizz, then the balloon will start to inflate!That lift is the gas produced from the two ingredients is carbon dioxide or CO212/5/18 · The reaction between the baking soda and vinegar cause the balloon to inflate all on its own!



Vinegar And Baking Soda Balloon Experiment Happy Brown House



Baking Soda And Vinegar Balloon Experiment Science Projects For Kids Educational Videos By Mocomi Youtube
Grab Baking Soda, Vinegar and a balloon and you're set to create an amazing gas called Carbon DioxiIf your seal isn't tight around the top of the bottle, your balloon may fly off like a rocket into the air, so watch out!26/11/12 · Gently shake the baking soda and vinegar mixture What happens?



At Home Lesson Breathless Balloons With Edie S Experiments Penguin Books Australia



Balloon Baking Soda Vinegar Science Experiment For Kids
18/11/ · Pour two teaspoons of baking soda into your balloon, and pour half a cup of acetic acid into the bottle An intense reaction will begin, releasing CO2 and making the balloon inflate If the reaction is too weak to inflate the balloon, add more vinegar and baking soda, but don't shake the solution7/6/ · Baking soda is bicarbonate (NaHCO3) and vinegar is acetic acid (HCH3COO) One of the products this reaction creates is carbon dioxide, which makes the bubbles When the baking soda meets the vinegar, there is a chemical reaction as carbon dioxide gas is created and fills the balloon causing it to inflateThis will allow the baking soda to drop out of the balloon and enter the flask Hold the bottle at the base while the reaction occurs The baking soda will quickly react with the vinegar in the flask, creating carbon dioxide gas as one of its products, causing the balloon to quickly inflate



Inflate A Balloon Using Chemistry In This At Home Experiment Wzzm13 Com



Wait Weight Don T Tell Me Chemistry Earth Science Science Activity Exploratorium Teacher Institute Project
Blowing Up a Balloon Science Experiment Explained Baking soda is a base and vinegar is an acid When the two materials mix, a chemical reaction occursThen, place the opening of the balloon around the opening of the bottle Try not to let any baking soda into the bottle as you do this Finally, when you are ready for some magic, lift the balloon to allow the baking soda to fall into the bottle As soon as the baking soda hits the vinegar, a chemical reaction takes place that produces carbonThe baking soda and the vinegar create an ACIDBASE reaction and the two chemicals work together to create a gas, (carbon dioxide) Gasses need a lot of room to spread out and the carbon dioxide starts to fill the bottle, and then moves into the balloon to inflate it



Baking Soda And Vinegar Balloon Experiment Youtube



Self Inflating Balloon Baking Soda And Vinegar Balloon Experiment Teach Beside Me
It is a showstopper experiment for kids (See more of my STEM projects for kids) How to Do the Baking Soda and Vinegar Balloon Experiment Supplies You Will Need for the ExperimentThe baking soda and vinegar will react together and begin to bubble and expand first Then you will see your balloon start to grow and grow until the reaction slows down and the balloons will begin to shrink again (or you can quickly take them off while they are full and tie them too) Baking soda and vinegar balloon experiment explanation5/6/18 · Today I want to show you Balloon Blow Up Vinegar VS Baking Soda Science Experiment Balloon Blow Up Vinegar VS Baking Soda Science ExperimentHello everyone!



Baking Soda Vinegar Balloon Experiment Awesome Pcl



Vinegar And Baking Soda Balloon Experiment Happy Brown House



Vinegar And Baking Soda Balloon Experiment Happy Brown House



Classroom Resources Inflating A Balloon With Chemistry ct



Blowing Up A Balloon With Baking Soda And Vinegar Cool



Baking Soda And Vinegar Balloons



Baking Soda And Vinegar Balloon Experiment Science Project Education Com


Science With Children Baking Soda And Vinegar Experiments



Balloon Science Experiment For Preschoolers From Abcs To Acts From Abcs To Acts



Baking Soda And Vinegar Balloon Experiment Science Stock Vector Image Art Alamy



Baking Soda And Vinegar Balloons



Koch Newsroom Baking Soda Balloons



Baking Soda And Vinegar Balloon Experiment



How To Inflate Balloon With Vinegar And Baking Soda Science4fun



How To Blow Up A Balloon With Baking Soda And Vinegar At Home Science Experiments For Kids Jm Cremp S Adventure Blog



Self Inflating Balloon Baking Soda And Vinegar Balloon Experiment Teach Beside Me



Museum At Home Diy Fizzy Balloon Monster Head The Children S Museum Of Indianapolis



Watercolor Balloons For Kids Learn Colors With Color Balloons Baking Soda And Vinegar Experiment Youtube



Vinegar And Baking Soda Balloon Experiment Happy Brown House



Magic Balloon Inflation Baking Soda And Vinegar Experiment Midwestern Mama



Baking Soda And Vinegar Balloon Experiment



States Of Matter Balloon Inflation Experiment Baking Soda Vinegar



How To Inflate Balloon With Vinegar And Baking Soda Science4fun



How To Fill A Balloon With Baking Soda Vinegar 5 Steps With Pictures Instructables



Blow Up Balloon This Time With Baking Soda And Vinegar Science And Samosa



Baking Soda Vinegar Balloon Experiment Easy Fun Kid Friendly Things To Do


Koch Newsroom Baking Soda Balloons



Balloon Blow Up Science Experiment



Simple Science Experiment Chemical Reaction Brigitte Brulz



How To Inflate A Balloon Using Baking Soda And Vinegar



Baking Soda Vinegar And Our Atmosphere Experiment Wltx Com



Giant Balloon Baking Soda And Vinegar Experiment Hello Wonderful



Blowing Up Giant Balloon Baking Soda And Vinegar Experiment For Kids Youtube


Baking Soda Vinegar Balloon Experiment Capturing Parenthood



Blowing Up Balloons With Baking Soda And Vinegar Pink Stripey Socks



How To Blow Up A Balloon With Vinegar And Baking Soda Or Yeast



Balloon Science Experiment For Preschoolers From Abcs To Acts From Abcs To Acts



Baking Soda And Vinegar Balloon Science Project Education Com



Balloon Baking Soda Vinegar Kids Science



Balloon Baking Soda Vinegar Experiment For Kids Bilingual Education Activities



Inflate A Balloon Career Girls



Baking Soda Vinegar Balloon Experiment The Go To List



Baking Soda And Vinegar Balloons



Blow Up A Balloon With Baking Soda And Vinegar Frugal Fun For Boys And Girls



Blow Up A Balloon With Baking Soda And Vinegar Frugal Fun For Boys And Girls Cool Science Experiments Science Fair Projects Fun Science



Baking Soda And Vinegar Balloons



Super Fun Experiment For Kids And Big Kids Blow Up Balloons With Vinegar Baking Soda These Will Not F Blowing Up Balloons Fun Experiments For Kids Balloons



Baking Soda And Vinegar Balloon Experiment For Kids Science Experiments Kids Kid Experiments Balloon Experiment



Balloon Blow Up Science Experiment



At Home Lesson Breathless Balloons With Edie S Experiments Penguin Books Australia



Baking Soda And Vinegar Reaction Earth Day Science Experiment


Science Experiments For Kids Blow Up A Balloon With Vinegar And Baking Soda



Baking Soda And Vinegar S Reaction Perkins Elearning



Self Inflating Balloon Science Experiment


Baking Soda Vinegar And A Balloon Oh My Scientific Method Data Sheet



Mama S Little Muse Science Activity Blowing Up A Balloon Using Baking Soda And Vinegar



Chemical Reaction Experiment When Baking Soda Stock Photo Edit Now



Science Project How To Inflate Up A Balloon With Liquid Ppt Video Online Download



Balloon Magic With Baking Soda And Vinegar Gift Of Curiosity



Baking Soda And Vinegar Balloon Experiment Science Royalty Free Cliparts Vectors And Stock Illustration Image



Balloon Magic With Baking Soda And Vinegar Gift Of Curiosity


Aswathi P



Balloon Baking Soda Vinegar Science Experiment For Kids



Baking Soda And Vinegar Balloons



Baking Soda Vinegar Balloon Experiment Youtube
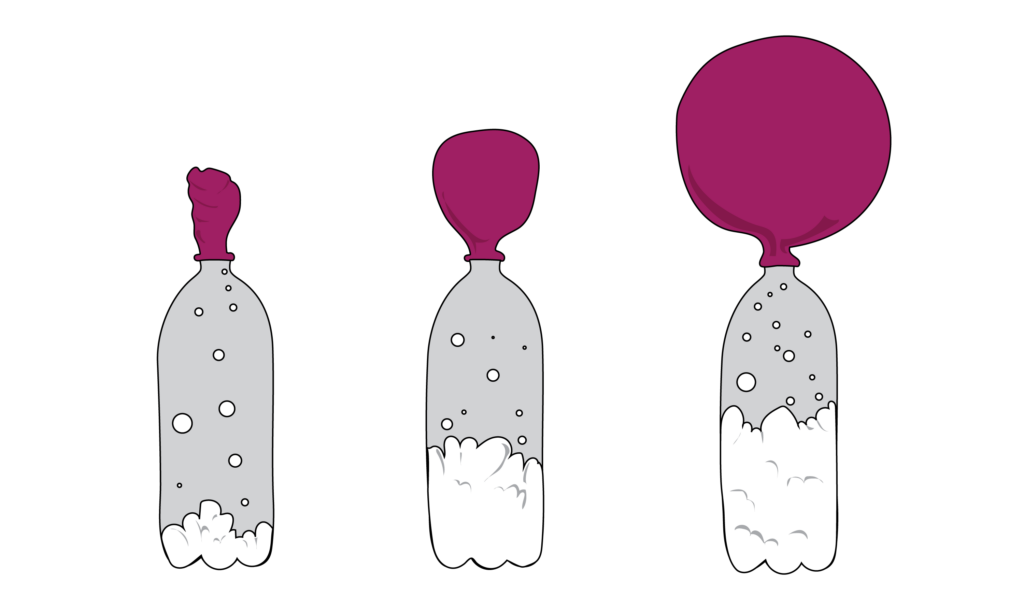


The Science Of Baking Soda Jerry James Stone



Baking Soda Vinegar Balloon Experiment Easy Fun Kid Friendly Things To Do



Baking Soda Vinegar And Our Atmosphere Experiment Wltx Com



Magic Balloons Playdough To Plato


Blowing Up Balloons With Co2 A Unique Hands On Science Night



Build A Fizz Inflator Sciencebob Com



Blow Up Balloon This Time With Baking Soda And Vinegar Science And Samosa



Self Inflating Balloon Baking Soda And Vinegar Balloon Experiment Teach Beside Me



Blowing Up A Balloon With Baking Soda And Vinegar Cool



Balloon Baking Soda Vinegar Kids Science



0 件のコメント:
コメントを投稿